Table of contents
Introduction
Efficient process design in modern energy, chemical, and manufacturing industries increasingly relies on accurate transport and management of thermal energy by engineered fluids. Understanding and quantifying heat capacity—essentially answering “How much heat can be transported by a fluid?”—is central for process engineers, R&D teams, scale-up managers, and quality specialists evaluating technology options for thermal management, system optimization, or specification compliance.
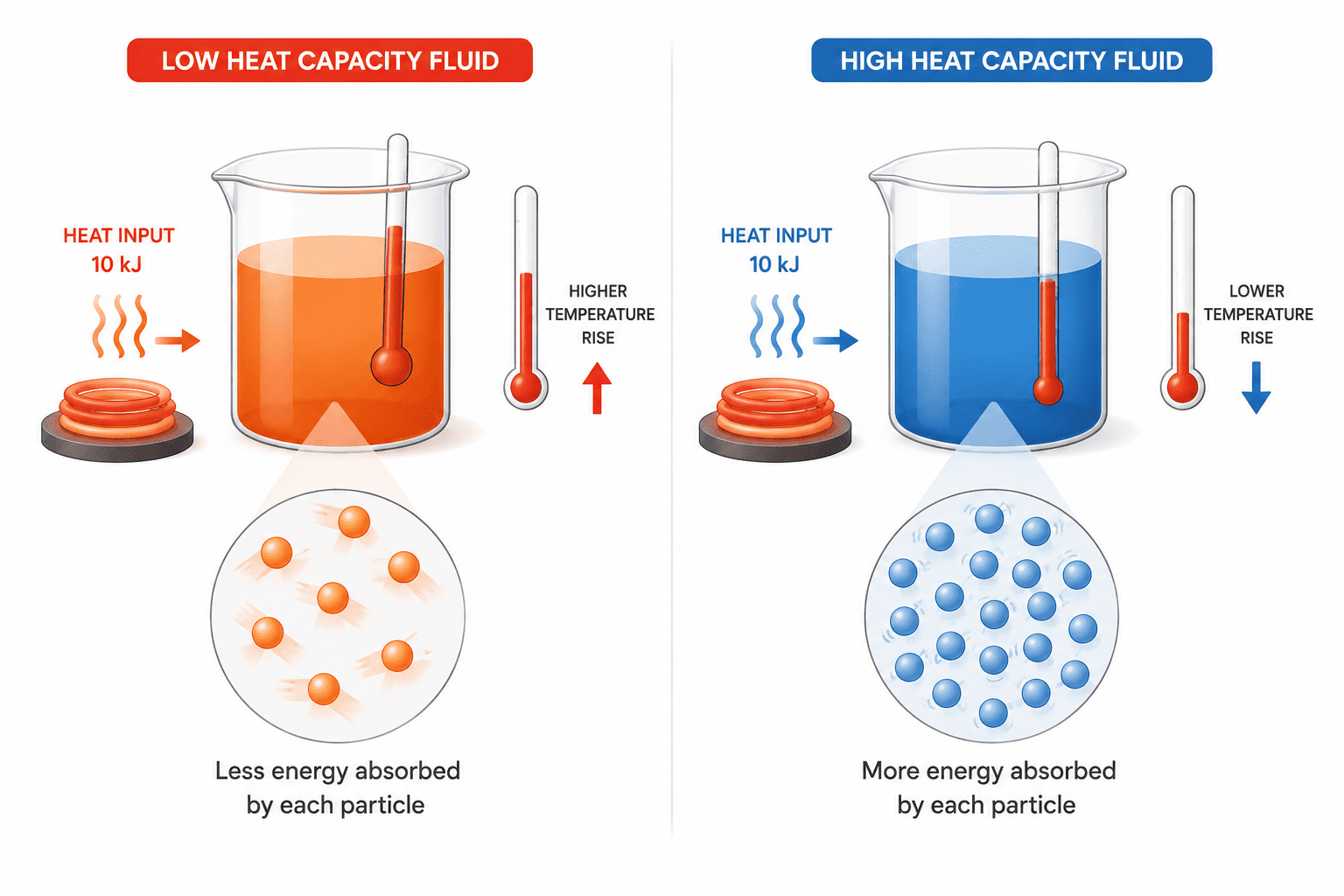
The Role of Heat Transfer Fluids in Process Design
Heat transfer fluids (HTFs) are used to collect, transport, store, and exchange thermal energy between system components. Applications range from large-scale chemical reactors and battery cooling systems to pilot plants and energy storage loops. Key requirements for HTFs include high heat capacity, stability across the intended temperature range, compatibility with construction materials, and safe operating profiles.
In process design, the heat that a fluid can transport (Q, in Joules) is determined by its mass (m), specific heat capacity (cp), and temperature change Q = mcpΔT.
Here, cp is the amount of heat required to raise the temperature of 1 kg of fluid by 1 K—a central parameter for HTF selection, system sizing, and energy balance calculations (Bauer, 2020).
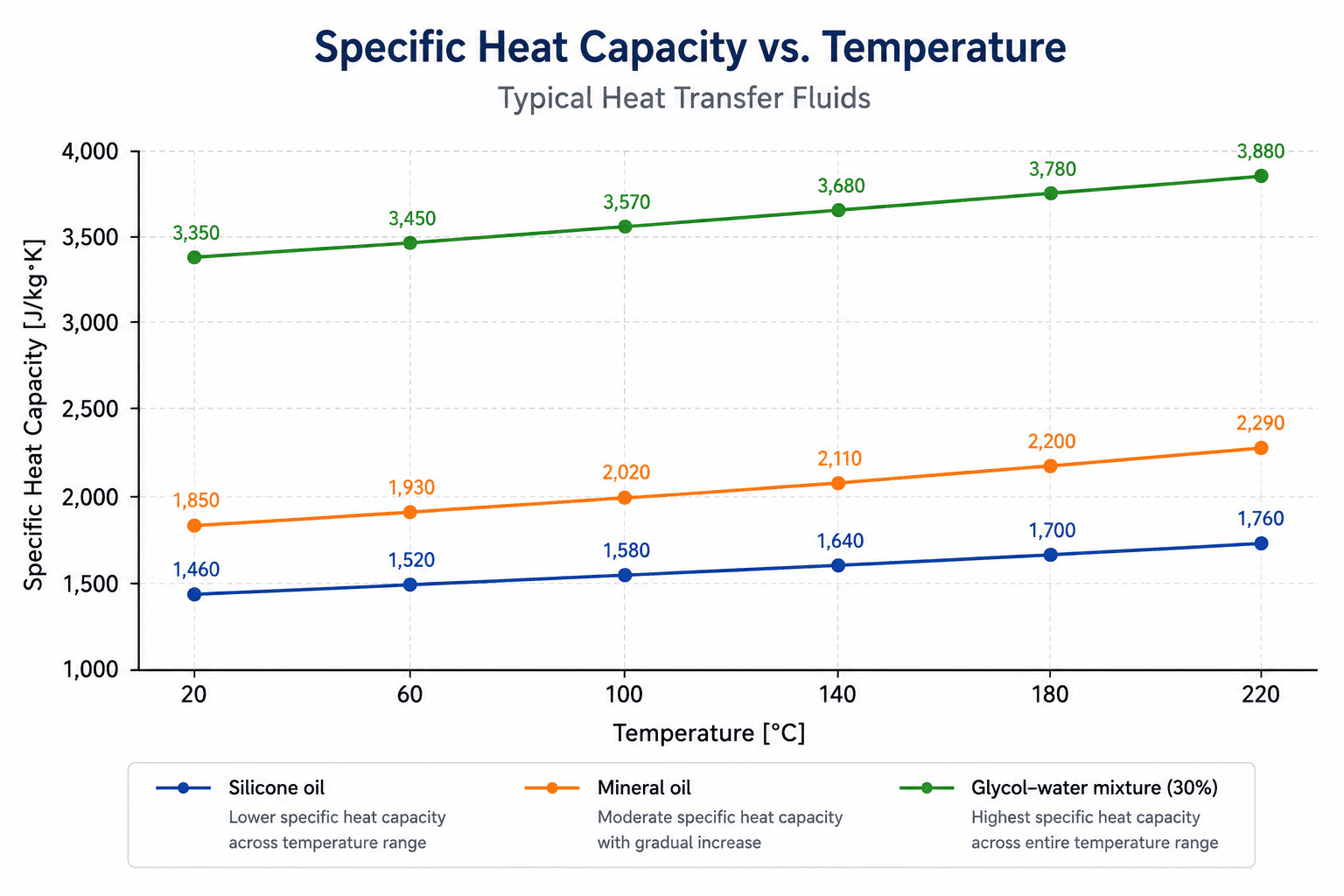
Illustrative values for visual explanation; exact values depend on fluid grade and measurement conditions.
Scientific Principles and Measurement Methods
Challenges in Accurate Heat Capacity Measurement
Material data sheets often provide generic values for HTF specific heat capacity, but real-world values can differ significantly from manufacturer data—especially after fluid aging, thermal cycling, or process contamination. Inaccurate heat capacity characterization can introduce substantial errors in efficiency estimation or thermal management performance (Lizana et al., 2018).
Two main approaches are used in practice:
Differential Scanning Calorimetry (DSC): A gold-standard method that directly measures the heat flow into a sample versus a reference under controlled temperature ramps. DSC is well-suited for laboratory analysis but may require adaptation for volatile or pressurized fluids at high temperatures. The volumetric heat capacity of non-porous solids and liquids is typically in the range 1.5 to 6 MJ·m⁻³·K⁻¹, with measurement uncertainties in DSC typically ranging between 2% for well-defined samples and up to 20% for complex or unstable fluids (Bauer, 2020). Process variables such as sample moisture, crucible integrity, and calibration significantly impact reliability.
Flow-Through Calorimetry: This technique operates under industrially relevant conditions, measuring heat capacity directly in operating loops. Studies have demonstrated that flow calorimeters can yield measurement uncertainties below 1.2% for HTFs at temperatures up to 330°C by combining precise temperature, mass flow, and electrical heating measurements. Validation measurements using water have revealed deviations below 0.1% from reference values at ambient temperatures, while field measurements at solar thermal installations showed deviations of under 1% from reference water measurements and up to 3.7% above 270°C for thermal oils. Notably, manufacturer specifications have been found to deviate by up to 10% from true values, highlighting the importance of on-site testing under real thermal stress and flow regimes (Bauer, 2020).
Advanced Fluid Concepts and Storage Enhancement
A novel trend in thermal fluids research is the development of reactive fluids whose heat capacity can be increased via reversible chemical reactions. Research has demonstrated that thermal fluids configured with specific reactants can surpass the heat capacity of water in certain temperature ranges, expanding both the operating window and the amount of heat transported per unit mass. Such approaches show promise for next-generation energy storage or high-performance cooling, with demonstrated increases in enthalpy storage of approximately 40% compared to conventional fluids over extended temperature windows (Lizana et al., 2018).
Fluid Degradation and Impact on Heat Transport Efficiency
Fluid degradation or contamination has a significant negative effect on both the heat capacity and heat transport efficiency of heat transfer fluids over time. Degradation is typically caused by prolonged exposure to high temperatures, thermal cracking, oxidation, and contamination from process leaks or reaction byproducts.
Impact Mechanisms
Reduction in Heat Capacity: As the fluid degrades, chemical changes directly alter the composition and thermal properties, often lowering the specific heat capacity and restricting the fluid’s ability to store and transport energy per unit mass (Bauer, 2020).
Thermal Transport Efficiency Loss: Degraded fluid tends to leave carbonaceous or polymeric residues on system surfaces, creating insulating layers. This fouling reduces the heat transfer coefficient, increases required energy input to maintain process temperatures, and causes higher operating costs and lower system efficiency.
Contamination Effects: Internal contamination can arise from ingress of process materials, water, or external substances, which further degrade fluid performance. This can lead to phase separation, unpredictable changes in viscosity and heat capacity, and accelerated corrosion or further fouling throughout the heat transfer loop.
Practical Consequences
Increased fouling leads to more frequent maintenance shutdowns and cleaning cycles, directly impacting productivity and process reliability. Severe degradation may result in fluid auto-ignition, hazardous vapor production, or equipment damage, especially as local temperatures at fluid film surfaces can rise above safe operating limits. Regular system monitoring for temperature excursions, chemical analysis of fluid samples, and timely replenishment or replacement of degraded fluid are key best practices to maintain heat transfer performance and avoid costly breakdowns.
Early Detection of Thermal Fluid Degradation
Early detection of thermal fluid degradation is critical for maintaining process efficiency and preventing costly downtime. Several measurable parameters provide early warning signs of fluid aging and breakdown, enabling proactive maintenance and replacement decisions.
Key Early Indicators
Acid Number (Total Acid Number, TAN): TAN rises due to oxidation and the formation of acidic degradation products. Frequent measurement is recommended; even a moderate increase signals advancing degradation and impending sludge/resin formation.
Viscosity: Significant increases (>30%) indicate polymerization, oxidation, or accumulation of high boilers. A drop in viscosity suggests presence of low boilers from thermal cracking; both changes impair heat transfer and safety.
Flash Point: Decreases as low boilers or volatile breakdown products appear, raising operational and safety risks.
Vapor Pressure: An elevated vapor pressure often reflects the accumulation of volatile low boilers, which can lead to cavitation and pump failures.
Physical Appearance: Darkening of fluid, appearance of particulates, or off-odors often signal physical and chemical aging.
Sampling Guidelines for Trend Analysis
For reliable trend analysis of thermal fluid degradation, samples should generally be taken every 3 to 6 months during normal operation, with increased frequency (every 1 to 3 months) in the first year of system startup, after fluid replacement, or in high-stress applications (Lizana et al., 2018). After process upsets or changes, additional sampling is recommended to catch accelerated degradation early. For critical or high-temperature applications, shorter intervals maximize early-detection reliability.
Implications for Technology Selection and Process Engineering
Key Decision Criteria
When selecting and qualifying heat transfer fluids for industrial processes, engineers must consider:
- Transported Heat Quantity: Determined by mass flow, heat capacity, and the allowable temperature excursion.
- Thermal Stability and Aging: Real HTF performance can change over time, influencing the amount of heat a system can reliably move. In-situ measurement is advised for critical applications.
- Measurement and Validation Techniques: Integrating both DSC lab data and flow calorimetry field data is best practice for establishing specification baselines and ensuring process safety and efficiency.
- Process Integration: The chosen HTF and its measured properties should be compatible with process materials, control systems, and safety protocols.
Case Studies and Practical Evidence
Solar Thermal Plants: Studies at solar thermal facilities have validated flow calorimetry under field conditions for high-temperature HTFs, demonstrating the technique’s capability to provide accurate measurements under real operating conditions (Bauer, 2020).
High-Performance Cooling: Research shows how molten salts can outperform oil-based HTFs in terms of total heat transport, though they require freeze protection, which impacts cost and practical operation.
Next-Generation Fluids: Thermochemical fluids with enhanced heat capacity demonstrate significant increases in enthalpy storage compared to conventional fluids, offering potential for pilot and scale-up projects targeting advanced energy systems (Lizana et al., 2018).
Conclusion: Scientific Best Practice for Heat Transport Fluid Evaluation
In process design, accurate quantification of the heat capacity of transfer fluids under relevant operating conditions is crucial for optimizing thermal system performance. Integrating laboratory DSC with in-situ flow calorimetry, while benchmarking against up-to-date research, enables engineers to make informed decisions on material specification, process qualification, and technology selection. Novel fluid formulations and advanced in-situ measurement methods continue to expand the options for energy systems, chemical production, and industrial-scale thermal management. Maintaining fluid integrity through regular monitoring and proactive management is essential for predictable heat capacity and efficient heat transport in industrial processes.
Bibliography
Bauer, T. (2020) ‘Fundamentals of high temperature thermal energy storage, transfer and conversion’, in Ultra-High Temperature Thermal Energy Storage, Transfer and Conversion. Woodhead Publishing Series in Energy, pp. 1-35. Available at: https://elib.dlr.de/138584/1/2020%20-%20Bauer%20-%20Chapter%201%20TES%20in%20UHTES.pdf
Lizana, J., Chacartegui, R., Barrios-Padura, A. and Valverde, J.M. (2018) ‘Advances in thermal energy storage materials and their applications towards zero energy buildings: A critical review’, Applied Energy, 203, pp. 219-239. Available at: https://core.ac.uk/download/pdf/157763138.pdf