Linear expansion coefficient
Linear expansion coefficient CTE – a measured variable in dilatometry
The expansion coefficients (linear expansion and volume expansion) are material properties. They are determined experimentally and given in mm/m*K.
Kelvin is the unit of measure for absolute temperature and for differences in Celsius scale temperatures.
Linear expansion coefficient
The linear expansion coefficient indicates by what length difference in relation to the total length L a solid body changes with a temperature change of one Kelvin.
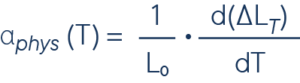
L0 = sample length, ΔLT = length change at temperature TT
The exact measurement of the linear expansion is analyzed with dilatometry. The samples are heated in an oven. The temperature profile follows a precisely predetermined program that allows the required heating rate, required temperature holding times and defined cooling processes. During this process the sample dimensions are continuously recorded. The different dilatometers of the Linseis are optimized for special tasks and equipped with extensive evaluation routines.
Coefficient of volume expansion:
The volume-specific coefficient of expansion indicates the amount by which the volume of a body increases or decreases in relation to the total volume when the temperature changes by one Kelvin.

Thermal expansion in daily life
Substances react to a change in temperature with the change in their volume.
This affects all areas of technology. In construction and in road, bridge and rail construction expansion joints must be planned, which absorb the longitudinal expansion of the building materials.
In pipelines expansion compensators in the form of built-in pipe bends are provided. Overhead lines for the power supply must be designed in such a way that the wires do not break in winter and have sufficient distance to the ground in summer.
The basis for the calculation of these and many other constructions is the thermal coefficient of linear expansion.
When storing liquids, a specified maximum filling level must be observed. Above the liquid level a space remains free, which absorbs the volume fluctuations. The calculation of such problems uses the volume expansion coefficient.
The terms derive from the fact that most substances expand when heated. With the few exceptions where substances contract when heated, the values of the two sizes are negative.
In the case of isotropic substances whose properties are independent of the direction considered, the volume expansion coefficient is three times the coefficient of linear expansion.
