Table of contents
The development of modern polymer blends focuses on the targeted combination of different polymers. polymers in order to flexibly adapt material properties to application requirements. Incompatibilities or phase separation within these blends represent one of the most common challenges, as they have a significant influence on the mechanical and thermal properties of the end product. Early identification of such incompatibilities is therefore crucial in order to ensure the quality, service life and performance of high-performance plastics.
The importance of phase separation in polymer blends
Phase separation refers to the separation of a polymer blend into two or more coexisting phases with different chemical or physical properties (Binder, 1987). This separation often results in degraded mechanical properties, such as reduced impact strength and toughness of the blend, and can be a critical failure mechanism, particularly in engineering applications. The effects range from visible surface defects to complete material failure under service conditions.
The compatibility of two polymers determines their miscibility: compatible blends show a homogeneous phase with uniform properties; incompatible systems, on the other hand, tend to segregate and form separate domains. This phase separation can occur at both macroscopic and nanoscopic levels, with the latter often only becoming apparent through specialized analytical methods.
The thermodynamic miscibility is described by the Gibbs mixing energy:
$$
\Delta G_{\mathrm{mix}} = \Delta H_{\mathrm{mix}} – T\,\Delta S_{\mathrm{mix}}
$$
For a spontaneous mixture, ΔG_mix must be negative. In polymer mixtures, the entropy term (TΔS_mix) is usually low due to the long chain molecules, so that the enthalpy of the mixture (ΔH_mix) is the decisive factor. The Flory-Huggins interaction parameter χ quantifies these interactions and enables predictions to be made about phase stability. Values of χ < 0.5 indicate compatibility, while higher values favor phase separation.
The critical temperature below which phase separation occurs can be determined experimentally and is essential for the definition of processing temperatures and application areas. Energy levels of the phases provide a relevant theoretical framework for estimating miscibility and have become established for the description and modeling of phase separation in practice.
DSC/DTA as a methodological key for laboratory practice
When screening polymer blends for compatibility, the differential thermal analysis (DTA) is a versatile, material-friendly method. DTA measures temperature differences between the sample and reference under controlled temperature conditions and is therefore ideal for detecting thermal events such as glass transitions, crystallization or melting processes.
The DTA uses a measurable temperature difference (ΔT = T_probe – T_reference) that occurs in the material during thermal treatment as a result of endothermic or exothermic processes. The practical implementation requires precise control of the experimental parameters: Typical heating rates are between 5 and 20 K/min, the sample amount between 5-20 mg, which makes DTA a material-friendly analysis method. An inert gas atmosphere prevents oxidative degradation of the polymer samples.
The differential scanning calorimetry (DSC, Differential Scanning Calorimetry) is a further development of DTA and is the most widely used thermal analysis method in polymer analysis today. While DTA only measures temperature differences between the sample and the reference, DSC also measures the associated heat flow (in mW or mJ/s) – it therefore provides not only qualitative but also quantitative information about thermal events. Depending on the design, a distinction is made between heat-flux DSC and power-compensation DSC: In heat-flux DSC, the sample and reference are heated in a common oven chamber and the temperature difference is converted into a heat flux via a calibrated thermal resistance; in power-compensation DSC, on the other hand, both are kept at the same temperature in separate micro-ovens, so that the power difference supplied directly represents the heat flux. The typical measurement parameters largely correspond to those of the DTA: heating rates of 5-20 K/min and sample quantities of 5-20 mg are common, whereby the DSC also reliably detects weaker thermal events – such as weakly pronounced glass transitions in polymer mixtures – due to its higher heat flow resolution.
Early detection of phase separation by DTA
Differential thermal analysis is particularly valuable for the early detection of phase separation in polymer blends, as it reliably detects thermal transitions and characteristic events in the material (Balhara et al., 2021). In the context of polymer blends, DTA provides specific indications of incompatibility and incipient phase separation even before macroscopic defects occur.
Characteristic indicators of incompatibility
Identification of multiple glass transitions (T_g): In compatible polymer blends, the DTA thermogram typically shows a single, intermediate glass transition. However, if two or more separate T_g peaks appear, this indicates the existence of multiple, individually separated phases. The Gordon-Taylor equation enables the theoretical calculation of the intermediate glass transition:
$$
T_{g,\text{blend}} = \frac{w_1 T_{g1} + k w_2 T_{g2}}{w_1 + k w_2}
$$
Deviations from this relationship indicate incompatibility.
Analysis of peak shapes and widths: A broadening or asymmetry of transition signals often indicates overlapping, not completely separated phases or incipient segregation processes. The half-width of glass transition peaks correlates with the homogeneity of the mixture – narrower peaks indicate more homogeneous systems.
Separate melting and crystallization temperatures: If several melting or crystallization peaks are visible, this results from the coexistence of different crystalline phases. The occurrence of separate peaks indicates the thermal independence of the individual phases. The crystallinity can be quantified by integrating the melting peaks.
Shift of the transition temperature: If the measured transition temperatures shift compared to those of the pure polymers, this may indicate an interaction effect or the co-presence of incompatible components.
Application examples from practice
Technical thermoplastics: For PC/ABS blends (polycarbonate/acrylonitrile-butadiene-styrene), DTA clearly shows the two characteristic glass transitions at around 110°C (ABS) and 150°C (PC) for incompatible blends. The addition of compatibilizers causes these to merge into a broad, intermediate peak.
Biocompatible systems: PLA/PCL blends (polylactide/polycaprolactone) for medical applications show characteristic crystallization and melting behaviour. DTA analysis enables the optimization of degradation kinetics for special biomedical applications.
High-performance plastics: PEEK/PEI blends (polyetheretherketone/polyetherimide) for aerospace applications require precise thermal characterization. DTA identifies optimal mixing ratios for maximum temperature stability.
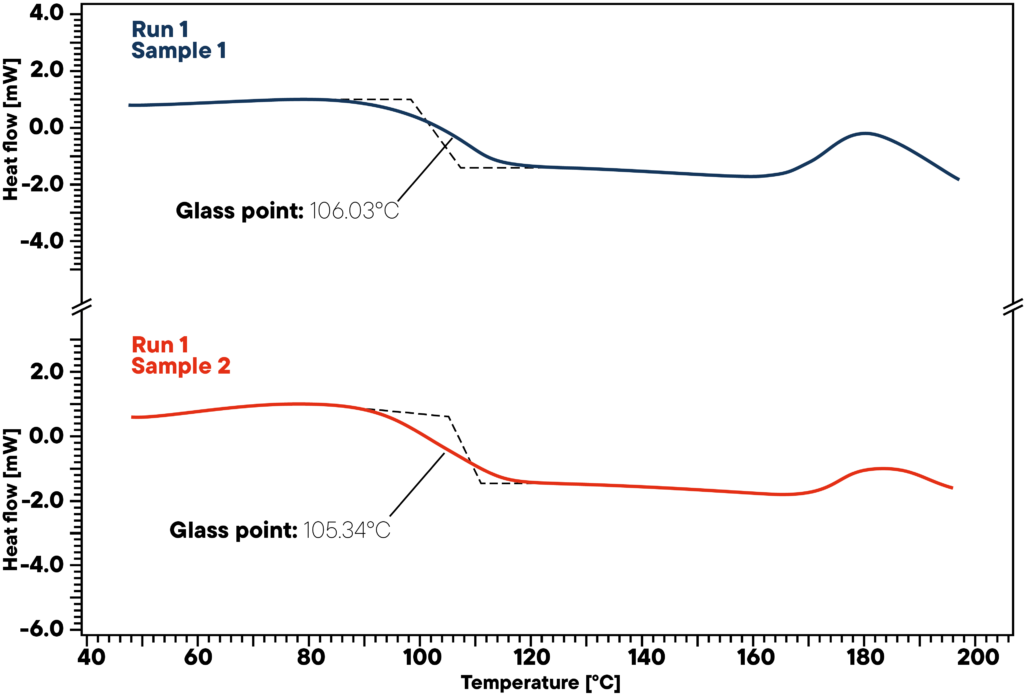
Thermal analysis is ideally suited to the investigation of polymers and polymer blends. In this measurement, two ABS samples were analyzed using differential thermal analysis (DTA) with the HDSC L62 was used. During a controlled temperature program, the temperature difference between the sample and the reference is recorded, making thermal transitions visible.
Both samples show the typical glass transition of ABS at around 105-106 °C. The almost identical transition temperatures of the two curves indicate a comparable material composition and quality of the tested samples.
Practical advantages and metrological aspects
The simple sample preparation, low material consumption and comparatively fast measurement make DTA a labor-saving method in polymer development. The quality of DTA measurements depends crucially on correct calibration – standard substances such as indium (melting point 156.6°C) are used for temperature calibration.
Early detection: The method provides clarity about incompatible mixtures at an early stage of development, thus avoiding misguided developments and cost-intensive iterations.
Material-friendly method: The analysis requires only small sample quantities (5-20 mg) and thus enables resource-saving characterization, even if the sample itself is altered in the course of thermal treatment.
Quality control: DTA shows phase separation or homogeneity even before they can be detected by the naked eye or mechanical testing.
Economic relevance: The early identification of incompatible formulations avoids costly pilot plant trials and can significantly reduce the development time for new developments.
Comparison with other characterization methods
Leading studies confirm the fundamental role of thermal analysis methods for the characterization of polymer blends. In scientific discourse, the occurrence of multiple glass transitions is considered an unmistakable sign of phase separation (Ivancic et al., 2024). Modern simulations use DTA data to validate theoretical predictions of the χ-parameter model.
Current trends are dedicated to more complex systems such as ternary blends and bio-based plastics. The latest research approaches combine DTA data with machine learning for automated classification of compatibility and prediction of optimal blend compositions.
The integration of DTA with other analysis methods is developing into hyphenated techniques: TGA-MS for simultaneous identification of degradation products or DTA-FTIR for the characterization of chemical changes during thermal transitions.
Standards and standardization
The standardization of DTA measurements follows internationally recognized standards: ASTM D3418 defines standard procedures for glass transition determinations, ISO 11357 describes DSC/DTA measurements on polymers in several parts – including glass transitions – and each has a counterpart in the ASTM series of standards. These standards ensure the comparability of measurement results between different laboratories and are mandatory for regulated industries.
Conclusion
Differential thermal analysis is an indispensable tool for the development of sustainable polymer blends. It allows phase separation and incompatibilities to be detected at an early stage, which is crucial for process and product optimization. As a robust method, DTA provides reliable thermal data that contributes to the targeted control of material development.
The thermally measurable signatures provide laboratory users and developers with quick indications of the microstructure and miscibility in the polymer blend. With the integration of artificial intelligence and miniaturization for fast laboratory measurements, DTA also promises to play a key role in polymer analysis in the future.
Bibliography
- Saxena et al. “Thermal analysis of polymer blends and double layer by DSC”, High Performance Polymers, 2021.
- Binder, K. “Dynamics of phase separation and critical phenomena in polymer mixtures”, Colloid and Polymer Science, 1987. DOI: https://doi.org/10.1007/BF01417926
- Kalogeras, I. M. “Glass-Transition Phenomena in Polymer Blends”, in: Encyclopedia of Polymer Blends, ed. A. I. Isayev, Wiley-VCH, 2016. DOI: https://doi.org/10.1002/9783527653966.ch1
- Ivancic, R.J.S. et al. “Predicting compatibilized polymer blend toughness”, Science Advances, 2024. DOI: https://doi.org/10.1126/sciadv.adk6165