Gypsum plaster is usually used at the outer surface and inner surface of brick walls to protect the bricks (or any other material the walls are made of) from weather and environmental influence.
The used gypsum is usually consisting of minerals, silicates and carbonates and of course water. It is mixed with water to a slurry and then applicated on the wall where it dries and hardens. The final product is a robust solid that should stay in shape for ages. Unfortunately, the exact procedure of mixing and the exact composition can have a huge influence on lifetime and robustness of the gypsum.
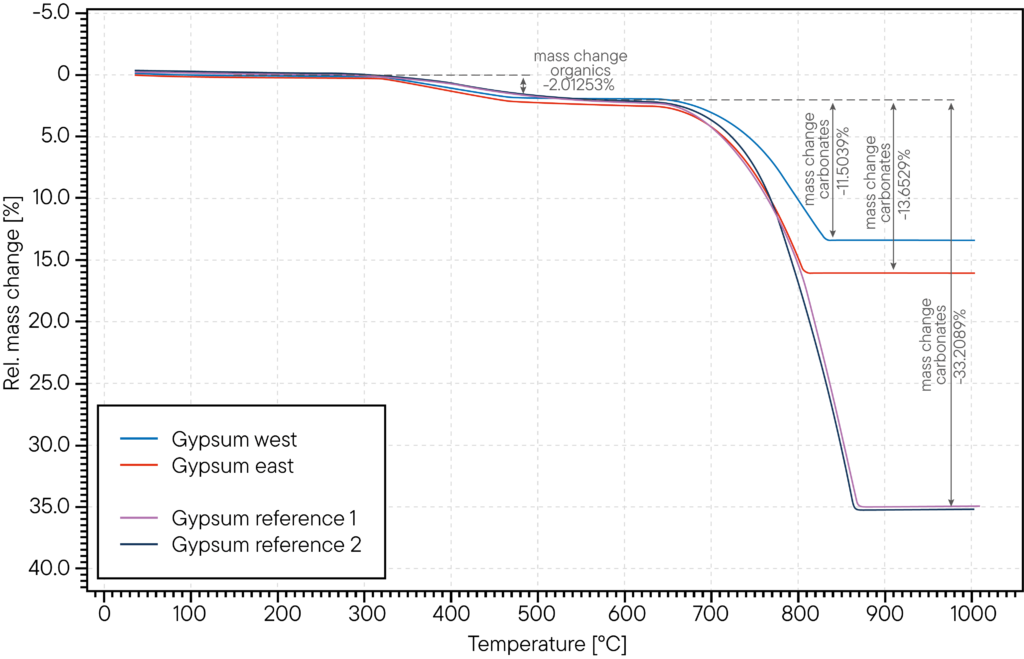
The measurement example below shows a damage analysis of a gypsum plaster that showed cracks and structural damage after a summer – winter cycle. The manufacturer assumed that the application was not performed properly and used thermogravimetric analysis (TGA) to compare the damaged wall part with a reference gypsum sample that did not show cracks after heating and cooling cycles. The measurement shows that the carbon and organic content of the “bad” samples (red and blue curves) is almost identical with the reference samples (purple and dark blue curve). The weight loss percentage shows the same level around 2 % mass loss in the range around 500 °C. However, there is a significant difference in the weight loss step around 800 °C – 900°C, where the contained carbonates are released as carbon dioxide: The reference samples show a mass loss of around 30 % caused by released CO2 while the samples from the wall with cracks show only 11 % mass loss and 13 % mass loss here. This indicates that the gypsum at the wall that showed structural failures has a significant lower carbon content than it should have which indicates that indeed the mixing of the gypsum plaster was performed wrong. Interestingly, there is also a difference in carbon content between the wall at the west side (weather facing side) and the east side of the building.
